RESEARCH
Research Interests
Every day, many different types of DNA damage threaten the integrity of our genome. A failure to properly repair these alterations can result in the acquisition of hallmarks of cancer such as translocations and somatic mutations, or can lead to cell death. In healthy cells, genomic stability is safeguarded by multiple sophisticated responses that specifically detect, signal and repair the different kinds of damage.
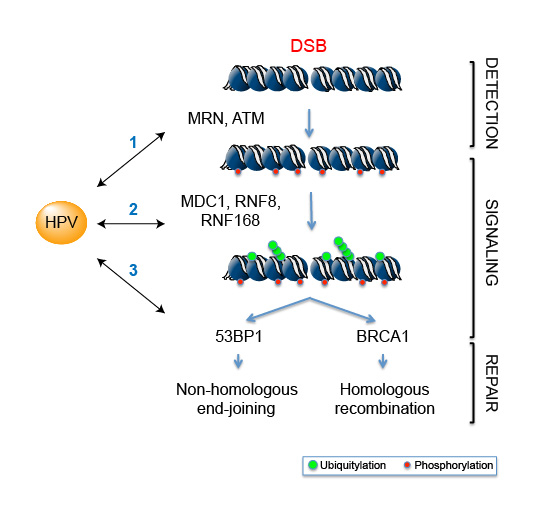
The main interest of my laboratory is to understand how these pathways are challenged during viral infections. We use a combination of molecular biology, biochemistry and cellular biology to determine how the infection impacts the genomic integrity of the infected cell and to tackle the mechanisms used by the human papillomavirus (HPV) to usurp the DNA damage machinery in order to promote the viral life cycle. Recently, we found that DNA double-strand break (DSB) signaling relies on the ability of specific proteins to interact with ubiquitylated chromatin and one of the main focuses of our work is aimed at understanding how these chromatin readers are affected upon viral infection. Specifically:
1. What is the interplay between HPV and the DSB signaling and repair proteins, and how does it change during carcinogenesis?
2. How does HPV impact the functions of the readers of ubiquitylated chromatin in cancer cells?
3. What are the consequences of viral infection on DSB signaling and DNA repair pathway choice?
As HPV is the causal agent of cervical cancer as well as of a subset of cancers of the head & neck and of the anogenital tract, the outcome of these studies will have important implications for HPV-dependent cancer biology as well as for our understanding of the viral life cycle. Opportunities are available for trainees interested in investigating the biology of HPV-positive cancer cells and in uncovering the molecular mechanisms used by the virus to hijack DNA damage responses in mammalian cells.
We are always looking forward to recruiting Masters and/or PhD students. If you are enthusiastic about training opportunities in the processes that regulate genomic stability during viral infection, please send us a research statement and your CV.


